
A
ires
FT
et
al
.
246
R
ev
A
ssoc
M
ed
B
ras
2016; 62(3):243-247
So far, there is no evidence to defend the prophylac-
tic use of lactulose in cirrhotic patients presenting trig-
gers for HE.
15
Based on the reasoning of the pathophysi-
ology of HE and the potential benefits of lactulose, the
clinical trials included in this review used lactulose pro-
phylactically to reduce the incidence of HE.
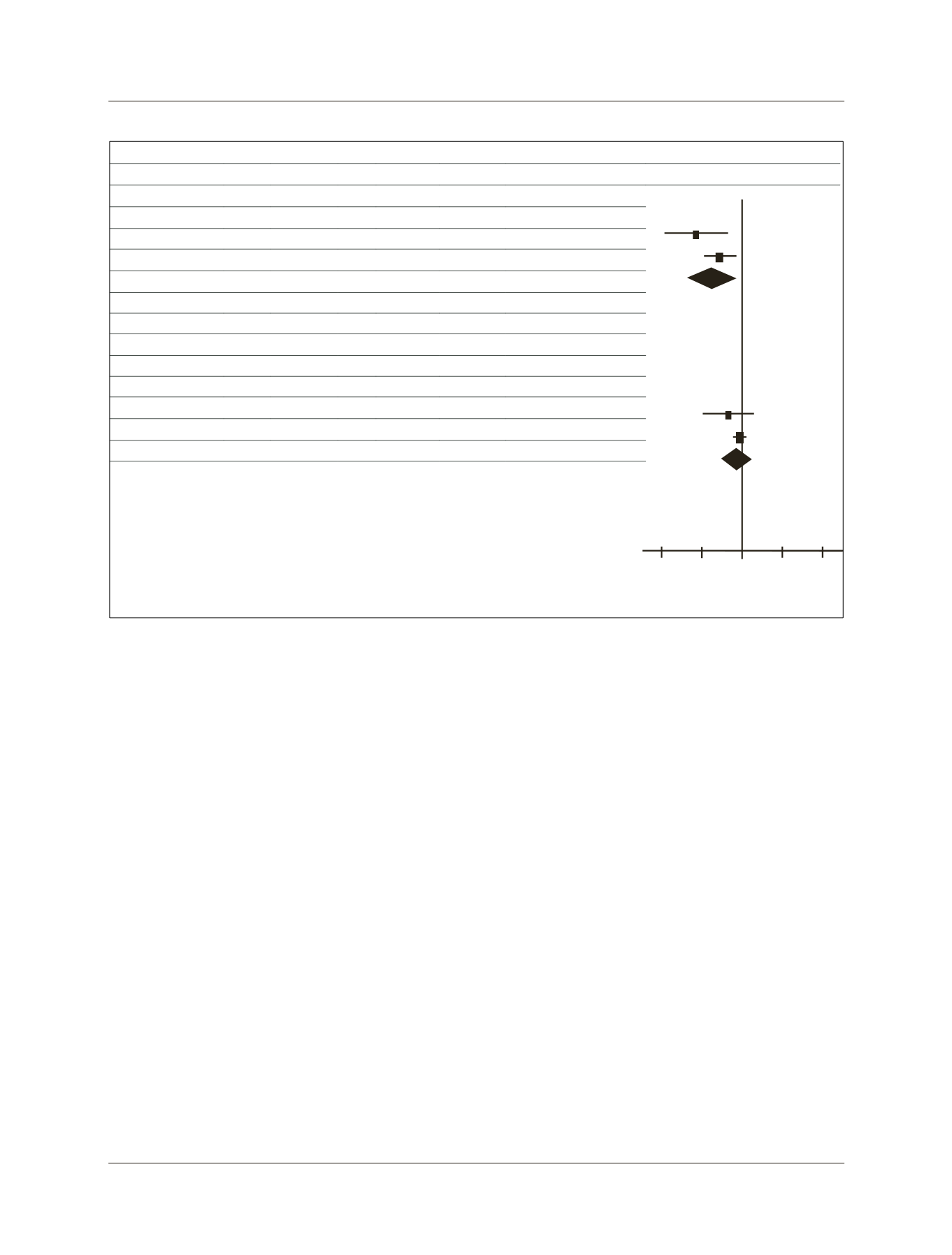
Both had a significant reduction in the incidence of
HE. Meta-analysis of the data showed a decrease of ap-
proximately 20% (range 10 to 28%) in the absolute risk
HE. The difference in incidence of HE between the pri-
mary studies can be explained by the fact that in the study
by Sharma et al. patients had higher Child-Pugh scores
(9
vs.
6 points), which means worst liver performance. In
addition, the authors included only patients with gastro-
intestinal bleeding of varicose source, which is known to
occur when the liver function is already considerably com-
promised. Another difference observed was the etiology
of liver disease. In the study by Sharma et al., alcoholic
etiology prevailed, while in the study by Wen et al.
9
the
main cause of liver disease was chronic infection with
hepatitis B virus.
There was no difference in the overall incidence of
mortality. The highest number of deaths observed in the
study by Sharma et al. is explained by the same reasons
mentioned above.
The most common adverse events arising from the ad-
ministration of lactulose are diarrhea, abdominal cramps,
nausea and flatulence. Although these side effects are com-
mon, they seldom lead to discontinuation of treatment.
It should be noted that the summary of the evidence
in this review may be biased by the low statistical power
of the sample in the primary studies.
C
onclusion
Administering lactulose to cirrhotic patients with upper
gastrointestinal bleeding reduces the incidence of hepat-
ic encephalopathy.
R
esumo
Eficácia da lactulose na profilaxia de encefalopatia he-
pática em pacientes cirróticos apresentando hemorragia
digestiva
Introdução:
encefalopatia hepática (EH) é fator de mau
prognóstico no paciente com cirrose hepática e sua inci-
Study
Lactulose
Control
Difference in risk
Difference in risk
n
Total
n Total
Weight
M-H, Random, 95CI M-H, Random, 95CI
Incidence of hepatic encephalopathy
-0.5
0.5
-0.25
Control
Lactulose
0.25
0
Sharma P, 2001 5
35
15 35
20.1% -0.29 [-0.49, -0.08]
Wen J, 2013
2
65
11 65
26.9% -0.14 [-0.24, -0.04]
Total (95CI)
100
100
47.1% -0.19 [-0.33, -0.04]
Total of events 7
26
Heterogeneity: Tau
2
= 0.01; Chi
2
= 1.89; df = 1 (p = 0.17); I
2
= 47%
Overall effect test: Z = 2.53 (p = 0.01)
Mortality
Sharma P, 2001
3
35
6 35
23.3% -0.09 [-0.24, 0.07]
Wen J, 2013
0
65
1 65
29.7% -0.02 [-0.06, 0.03]
Total (95CI)
100
100
52.9% -0.03 [-0.12, 0.06]
Total of events 3
7
Heterogeneity: Tau
2
= 0.00; Chi
2
= 1.68; df = 1 (p = 0.20); I
2
= 40%
Overall effect test: Z = 0.71 (p = 0.48)
FIGURE 1
Meta-analysis of the incidence of hepatic encephalopathy and mortality.















