
R
osa
MI
et
al
.
658
R
ev
A
ssoc
M
ed
B
ras
2017; 63(7):656-661
Killick et al.
13
showed unclear risk of bias to the reference
standard (prostate biopsy), since it is unclear whether all
participants underwent prostate biopsy, flow and time
(patient flow and time between the completion of the
EN2 test and biopsy). These criteria resulted in a high
risk of bias for the reference standard, with respect to
applicability criteria.
EN2 test vs. biopsy
The two studies had a combined sensitivity (Figure 3A)
of 66% (95CI 56-75) and a combined specificity (Figure
3B) of 89% (95CI 86-92). The DOR (Figure 3C) was 15.082
(95CI 8.432-26.977).
Begg’s funnel plot and Egger’s test were not performed
to assess the publication bias of the literature in all com-
parison models since only two studies were included.
D
iscussion
PCa is becoming a public health concern worldwide and PSA
test is not being recommended by its own creator, Profes-
sor Richard J. Ablin, who always say “PSA testing cannot
detect prostate cancer.” This is the first systematic review
TABLE 1
Characteristics of primary diagnostic studies on prostate cancer measuring urinary levels of EN2.
Author/
Year
Mean age Age
control
Design and
settings
N
Control
N PCa Sensibility
(%)
Specificity
(%)
TP FP FN TN EN2 cut-off
(µg/L)
Morgan
et al.
7
67 (44-83) 63 (42-86) Case-control
102
82
66
88.2
54 12 28 90 42.5
Killick et al.
13
53 (40-69) 54.3
(40-69)
Cross-sectional 392
21
66.7
89.3
14 42 7 350 42.5
PCa: prostate cancer; TP: true positive; FP: false positive; FN: false negative; TN: true negative.
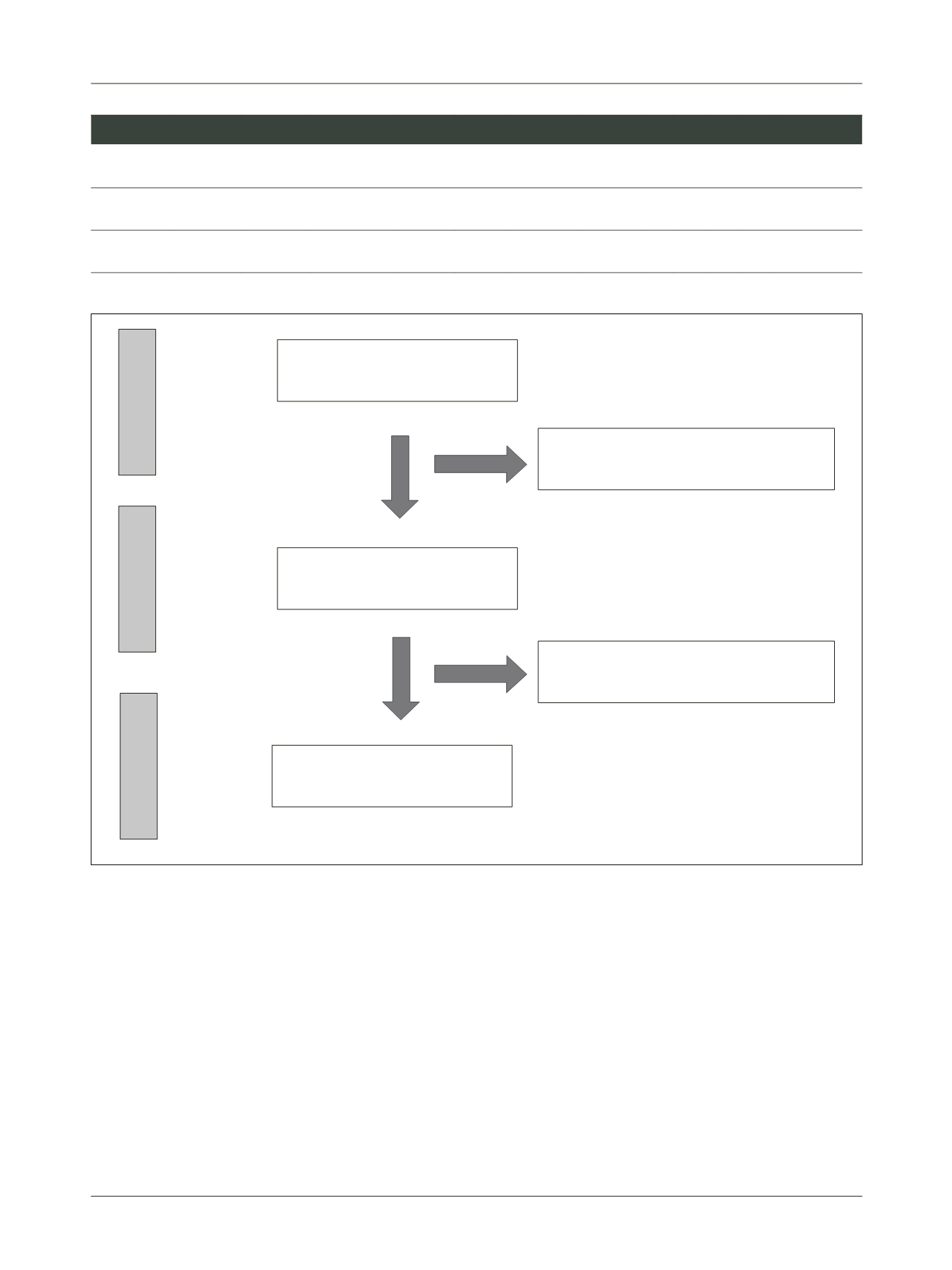
FIGURE 1
Flow diagram of the study selection process.
Identified through records database
(n=248)
Full text articles evaluated for eligibility
(n=17)
Studies that did not meet the inclusion criteria
based on title/abstract (n=231)
Full papers excluded, with reasons (n=15)
Studies included in the quantitative
synthesis (meta-analysis) (n=2)
Identification
Eligibility
Include
















